Haloalkanes and haloarenes form an important part of the Class 12 Chemistry syllabus, especially for board exams and competitive tests. This chapter focuses on organic compounds in which one or more hydrogen atoms are replaced by halogens like fluorine, chlorine, bromine, or iodine. The PDF you shared covers everything from basic definitions and classification to reaction mechanisms, stereochemistry, and real-life applications. It lays a strong foundation for understanding how halogen-containing compounds behave in different chemical environments.
I am writing about this chapter because many students find haloalkanes and haloarenes confusing due to the number of reactions, mechanisms, and exceptions involved. However, once the concepts are broken down logically, this chapter becomes very scoring. It also connects organic chemistry to real-world compounds like chloroform, DDT, and freons, making it important not just for exams but also for understanding environmental and industrial chemistry.
Introduction to Haloalkanes and Haloarenes
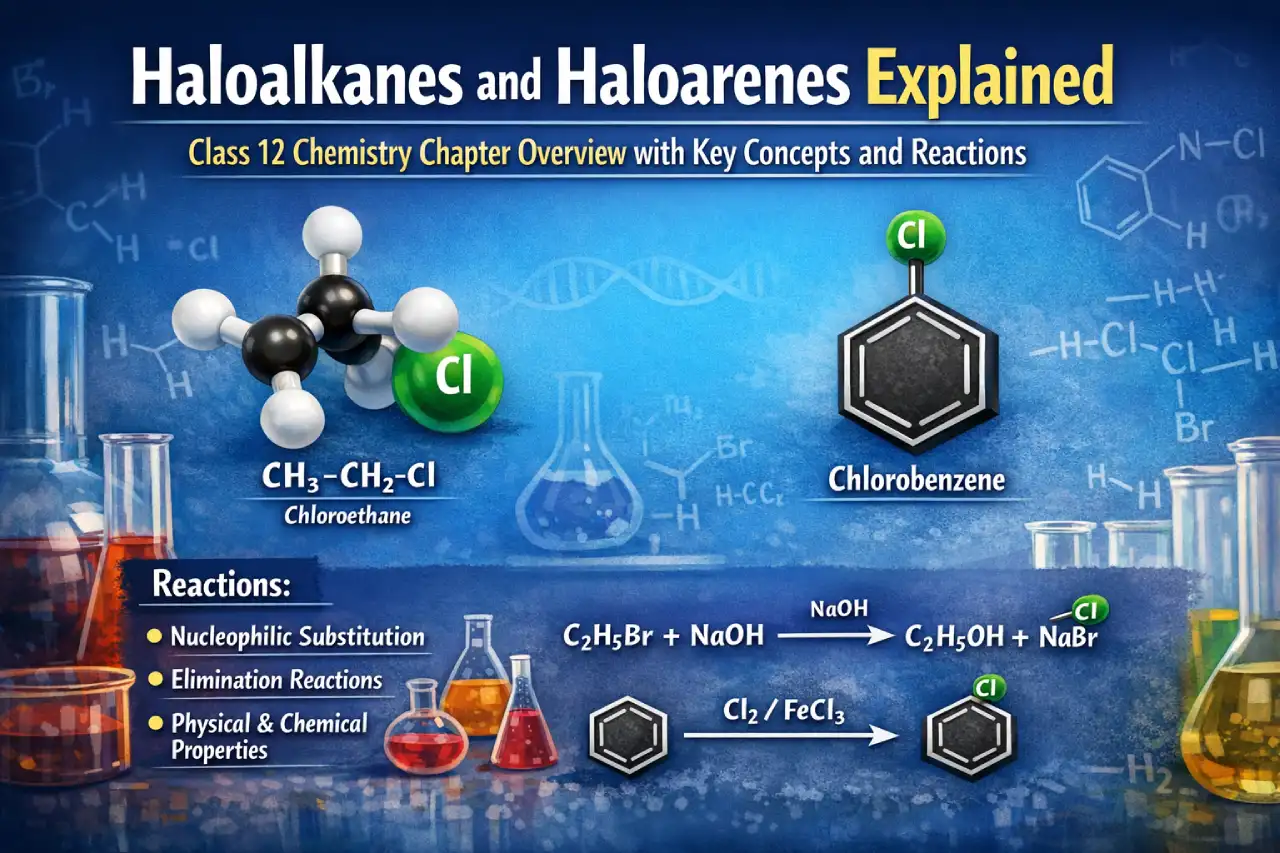
Haloalkanes are aliphatic hydrocarbons in which a halogen atom is attached to an sp³ hybridised carbon atom, while haloarenes are aromatic compounds where the halogen is directly attached to an sp² hybridised carbon of a benzene ring. The PDF clearly distinguishes between these two based on structure and bonding, which later explains why their chemical behaviour is different.
Classification of Halogen Compounds
The chapter classifies halogen compounds in multiple ways. Based on the number of halogen atoms, they are grouped as mono-, di-, or polyhalogen compounds. Dihalogen compounds are further divided into geminal and vicinal dihalides. Another important classification is based on hybridisation, which includes alkyl halides, allylic halides, benzylic halides, vinylic halides, and aryl halides. This classification helps in predicting reactivity and reaction mechanisms.
Nomenclature of Haloalkanes and Haloarenes
The PDF provides both common names and IUPAC names for a wide range of compounds. Tables list examples like chloromethane, chloroform, carbon tetrachloride, and benzyl chloride. Understanding nomenclature is essential because many exam questions directly test naming and identification of structures.
Methods of Preparation of Haloalkanes
Haloalkanes can be prepared using alcohols, hydrocarbons, and alkenes. The chapter explains reactions such as the use of HX, PCl₅, SOCl₂, and free radical halogenation. Addition reactions of alkenes follow Markovnikov’s rule, with anti-Markovnikov addition explained using peroxide effect. Halide exchange reactions like Finkelstein and Swarts reactions are also discussed in detail.
Physical Properties of Haloalkanes
The physical properties discussed include boiling point, melting point, density, and solubility. Haloalkanes generally have higher boiling points than corresponding hydrocarbons due to dipole–dipole interactions. The PDF clearly explains trends such as the increase in boiling point from chlorides to iodides and the effect of branching on boiling point.
Download this CLASS 12 – HALOALKANES AND HALOARENES PDF File: Click Here
Chemical Properties and Reaction Mechanisms
Chemical reactions of haloalkanes mainly depend on the cleavage of the carbon–halogen bond. The chapter explains nucleophilic substitution reactions (SN1 and SN2), elimination reactions, reactions with metals, and reduction reactions. The difference between SN1 and SN2 mechanisms is explained using carbocation stability, reaction order, solvent effects, and stereochemical outcomes like Walden inversion and racemisation.
Stereochemistry and Optical Activity
A major highlight of the PDF is the detailed explanation of stereochemistry. Concepts such as chiral carbon, enantiomers, diastereoisomers, racemic mixtures, optical rotation, and specific rotation are covered. Instruments like the polarimeter and principles like plane-polarised light are also explained to help students understand optical activity practically and theoretically.
Haloarenes: Structure and Reactivity
Haloarenes differ from haloalkanes because of resonance and partial double bond character in the C–X bond. This makes nucleophilic substitution difficult. The chapter explains why haloarenes undergo electrophilic substitution mainly at ortho and para positions and discusses reactions like Dow’s process, Sandmeyer reactions, and reactions with metals CLASS 12 – HALOALKANES AND HALO….
Polyhalogen Compounds and Their Uses
The PDF also covers important polyhalogen compounds such as dichloromethane, chloroform, iodoform, carbon tetrachloride, freons, and DDT. Their methods of preparation, properties, uses, and environmental impact are explained. Special attention is given to the harmful effects of stable compounds like DDT and why they are banned in many countries.
NCERT-Based Questions and Exam Focus
Towards the end, the chapter includes NCERT in-text and exercise questions with detailed answers. These cover naming, reaction predictions, mechanism-based reasoning, and conceptual explanations. This section is especially useful for exam preparation as it reflects the exact pattern of board-level questions.