Organic compounds containing oxygen form one of the most important sections of organic chemistry, especially for senior secondary and competitive exam preparation. This topic mainly deals with how oxygen is present in organic molecules in different functional groups like alcohols, phenols, ethers, aldehydes, ketones and carboxylic acids. The PDF explains their classification, structure, methods of preparation, physical properties and key chemical reactions in a systematic way, supported by examples and reaction mechanisms.
I am writing about this topic because it appears repeatedly in board exams, NEET, and other entrance tests, and many students struggle to connect reactions with concepts. Understanding this chapter properly helps in solving numerical problems, reaction-based questions and assertion–reason type questions. This PDF is useful because it brings together theory, reactions and previous year questions in one place, making revision more practical and focused.
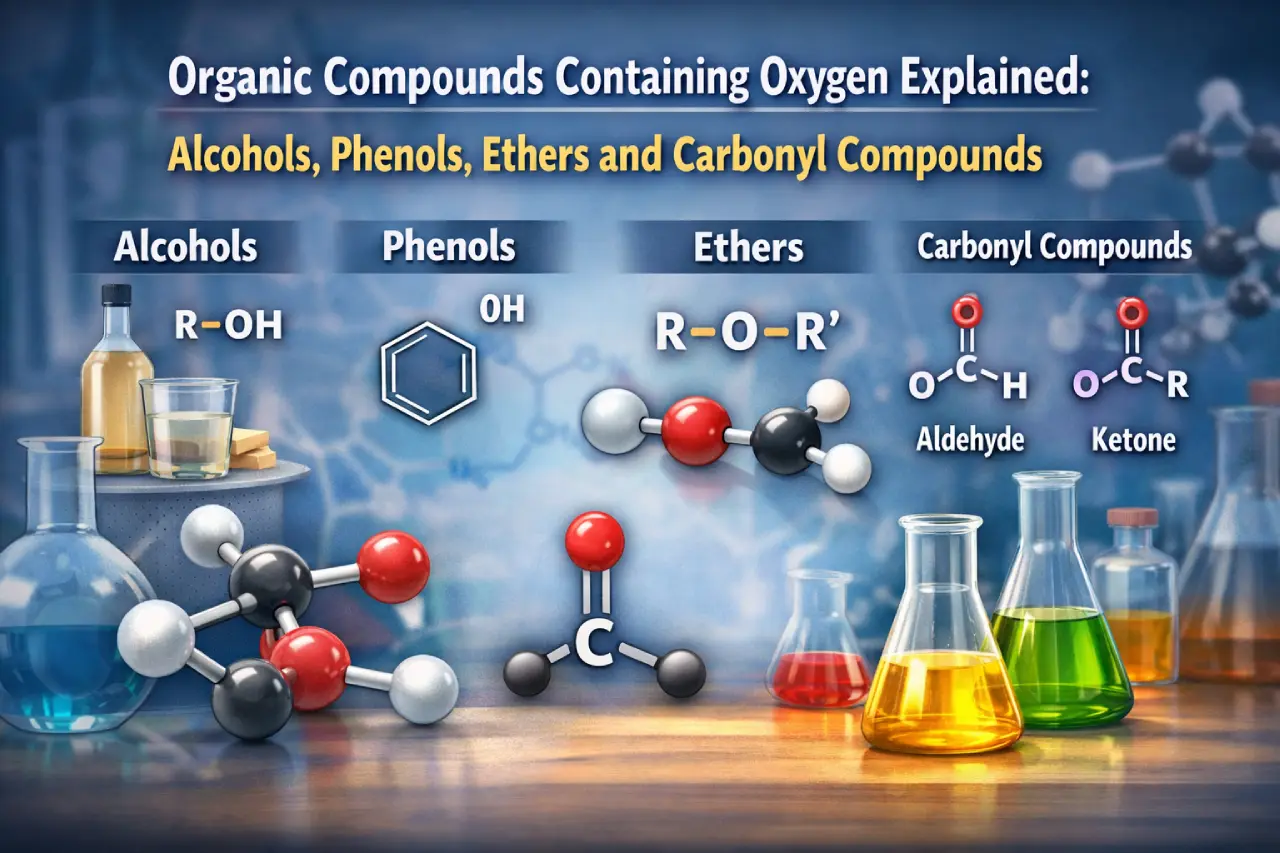
Alcohols and Their Classification
The PDF begins with alcohols, defined as compounds in which a hydroxyl group replaces a hydrogen atom of an alkane. Alcohols are classified based on the number of –OH groups as monohydric and polyhydric alcohols, and also as primary, secondary and tertiary alcohols depending on the carbon atom attached to the hydroxyl group.
It explains nomenclature using both common and IUPAC systems and includes common examples like methanol, ethanol, ethylene glycol and glycerol. Structural features and hydrogen bonding are clearly discussed to explain solubility and boiling point trends.
Preparation and Reactions of Alcohols
Various preparation methods are covered, including preparation from haloalkanes, alkenes, Grignard reagents, aldehydes, ketones, acids and esters. Each method is explained using chemical equations.
The reaction section focuses on substitution, oxidation, dehydration and esterification reactions. Special attention is given to Lucas test, iodoform test and oxidation reactions to distinguish between primary, secondary and tertiary alcohols.
Download this ORGANIC COMPOUND CONTAINING NITROGEN PDF File: Click Here
Phenols: Structure, Preparation and Chemical Behaviour
Phenols are described as compounds where the hydroxyl group is directly attached to an aromatic ring. The PDF explains classification into mono- and polyhydric phenols along with their nomenclature.
Methods of preparation include cumene process, diazonium salts, Dow’s process and Grignard reagent method. The acidic nature of phenol is explained using resonance stabilisation, followed by electrophilic substitution reactions such as halogenation, nitration and sulphonation.
Ethers and Their Properties
Ethers are introduced as compounds with the general formula R–O–R’. The PDF explains symmetrical and unsymmetrical ethers along with common and IUPAC naming methods.
Preparation methods like Williamson synthesis, dehydration of alcohols and alkoxymercuration are discussed. Physical properties such as low boiling points and limited water solubility are linked to the absence of hydrogen bonding. Cleavage of ethers using hydrogen halides is also explained.
Aldehydes and Ketones
This section explains the carbonyl group and its polarity. Aldehydes and ketones are differentiated based on structure and naming conventions.
Methods of preparation include oxidation of alcohols, ozonolysis, hydration of alkynes, Rosenmund reduction and reactions involving nitriles. Important reactions like nucleophilic addition, aldol condensation, Cannizzaro reaction, reduction and oxidation are explained with equations.
Carboxylic Acids and Their Derivatives
Carboxylic acids are introduced with their functional group structure and IUPAC naming rules. The PDF explains preparation methods from alcohols, aldehydes, nitriles, esters and Grignard reagents.
Physical properties such as acidity, boiling points and solubility are discussed using hydrogen bonding and inductive effects. Reactions like esterification, decarboxylation, reduction and substitution reactions are also included.
Importance of Reaction-Based Questions and Exam Focus
A large portion of the PDF is dedicated to previous year NEET and AIPMT questions. These questions help students understand how theory is tested in competitive exams.
Reaction mechanisms, identification tests and assertion–reason questions make this chapter scoring if studied properly.