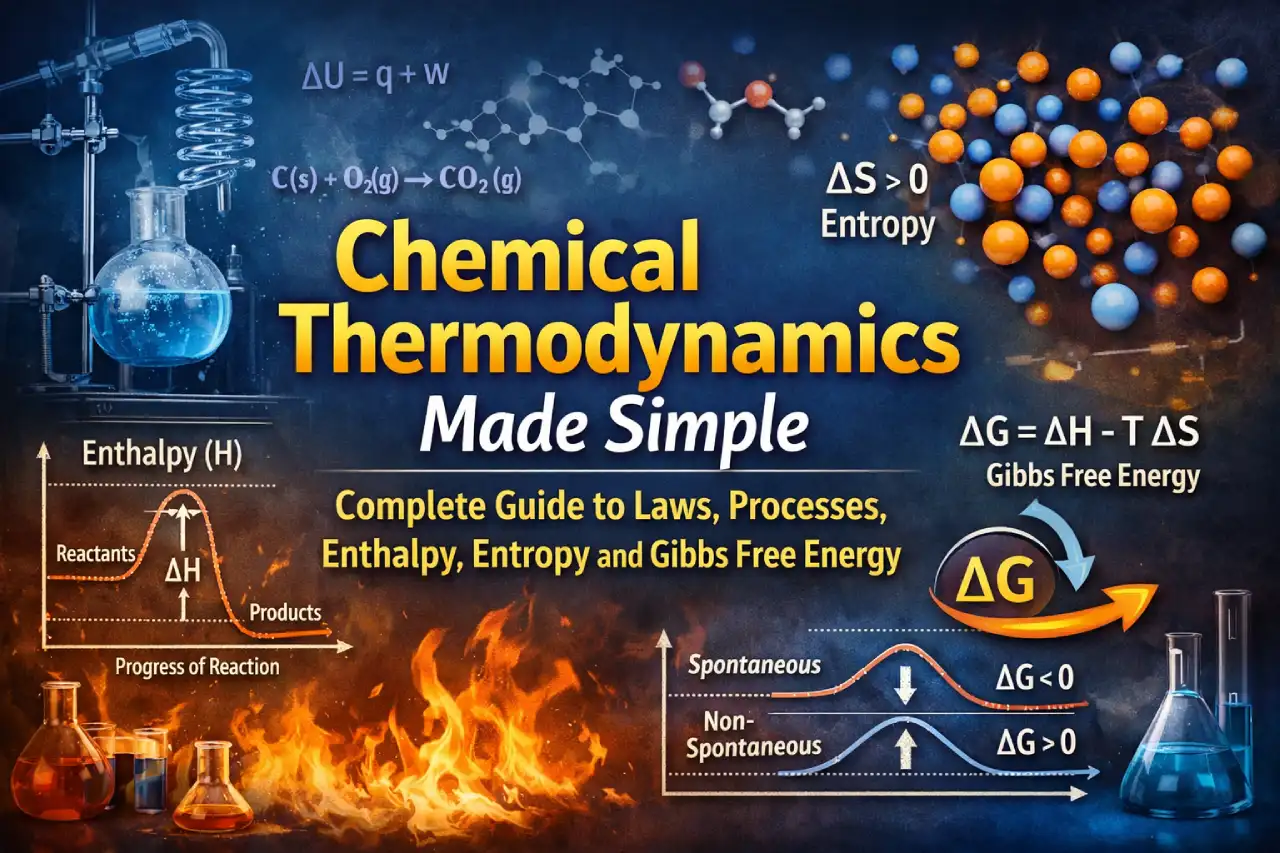
Chemical thermodynamics is one of the most important and scoring chapters in Class 11 Chemistry. It explains how energy changes take place during physical and chemical processes and helps us understand why some reactions occur naturally while others do not. The uploaded PDF on Chemical Thermodynamics covers all major concepts such as systems and surroundings, types of processes, laws of thermodynamics, enthalpy, entropy, Gibbs free energy, and their practical applications in a clear and structured way.
I am writing about this topic because many students find thermodynamics confusing due to formulas, sign conventions, and multiple laws. However, once the basics are clear, this chapter becomes logical and interesting. A strong understanding of chemical thermodynamics not only helps in board exams but is also extremely useful for competitive exams like JEE and NEET.
What Is Chemical Thermodynamics?
Chemical thermodynamics is the branch of science that studies the quantitative relationship between heat energy and other forms of energy during physical and chemical changes. It helps us predict:
- Whether a reaction is possible or not
- The amount of energy involved
- The extent to which a reaction can proceed
- The efficiency of engines and processes
Thermodynamics deals only with initial and final states, not with the speed or mechanism of reactions.
System, Surroundings and Universe
- System: The part of the universe under observation
- Surroundings: Everything outside the system
- Universe: System + Surroundings
Types of Systems
- Open system: Exchanges matter and energy
- Closed system: Exchanges only energy
- Isolated system: Exchanges neither matter nor energy
These classifications are essential for analysing energy flow in reactions.
Thermodynamic Properties
Intensive Properties
Do not depend on amount of matter
Examples: Temperature, pressure, density, pH
Extensive Properties
Depend on amount of matter
Examples: Mass, volume, internal energy, entropy, enthalpy
Ratio of two extensive properties always gives an intensive property, such as density = mass/volume.
Thermodynamic Processes
- Isothermal: Temperature constant
- Adiabatic: No heat exchange
- Isobaric: Pressure constant
- Isochoric: Volume constant
These processes describe how a system changes from one state to another.
Internal Energy (U)
Internal energy is the total energy stored in a system in different forms such as chemical, electrical, nuclear, and kinetic energy. Only the change in internal energy (ΔU) can be measured, not its absolute value.
First Law of Thermodynamics
Energy can neither be created nor destroyed, only converted from one form to another.
Mathematical form:
ΔU = q + W
Where
q = heat supplied
W = work done
This law establishes the principle of conservation of energy.
Enthalpy (H)
Enthalpy represents total heat content of a system.
H = U + PV
Change in enthalpy:
ΔH = ΔU + PΔV
Most chemical reactions in laboratories occur at constant pressure, so enthalpy change is widely used.
Heat Capacity
- Cv: Heat capacity at constant volume
- Cp: Heat capacity at constant pressure
For ideal gases:
Cp – Cv = R
This relation is very important for numerical problems.
Download this CLASS 11 – CHEMICAL THERMODYNAMICS PDF File: Click Here
Hess’s Law of Constant Heat Summation
The total enthalpy change of a reaction is the same whether it occurs in one step or several steps.
Applications:
- Determining enthalpy of formation
- Calculating lattice energy
- Finding heat of reactions not measurable directly.
Types of Enthalpy Changes
- Enthalpy of formation
- Enthalpy of combustion
- Enthalpy of neutralisation
- Enthalpy of solution
- Enthalpy of hydration
- Enthalpy of fusion
- Enthalpy of vaporisation
- Enthalpy of sublimation
Most combustion reactions are exothermic, while fusion and vaporisation are endothermic.
Second Law of Thermodynamics
All spontaneous processes are accompanied by an increase in entropy.
This law explains why certain processes occur naturally.
Entropy (S)
Entropy measures disorder or randomness of a system.
For a reversible process:
dS = δqrev / T
Criteria for spontaneity:
ΔStotal = ΔSsystem + ΔSsurroundings
If ΔStotal > 0, the process is spontaneous.
Gibbs Free Energy (G)
G = H – TS
Change in Gibbs free energy:
ΔG = ΔH – TΔS
Criteria:
- ΔG < 0 → Spontaneous
- ΔG = 0 → Equilibrium
- ΔG > 0 → Non-spontaneous
Gibbs free energy is the best criterion for predicting feasibility of reactions.
Relation Between ΔG° and Equilibrium Constant
ΔG° = –2.303 RT log K
This equation connects thermodynamics with chemical equilibrium.
Third Law of Thermodynamics
The entropy of a perfectly crystalline substance at absolute zero (0 K) is zero.
This law helps in calculating absolute entropies of substances.
Why Chemical Thermodynamics Matters for Students
- Builds strong conceptual base
- Important for physical chemistry topics
- Frequently asked in exams
- Helps in solving numerical problems confidently