Chemical bonding and molecular structure form the backbone of chemistry because they explain how matter is built at the atomic level. The uploaded PDF focuses on Chapter 4 of Class XI Chemistry and systematically introduces students to the forces that hold atoms together, the types of bonds formed, and the structural principles that govern molecules. From basic definitions like chemical bond and octet rule to advanced ideas such as molecular orbital theory and hydrogen bonding, the chapter builds a strong conceptual foundation.
I am writing about this PDF because this chapter is central to understanding almost every area of chemistry, whether it is organic reactions, inorganic structures, or physical properties of substances. Students often struggle with visualising molecules and linking theory with real behaviour. This PDF bridges that gap by combining explanations, formulae, examples, and practice questions in one place, making it an important resource for both board and competitive exam preparation.
Overview of Chemical Bonding and Molecular Structure
The PDF defines a chemical bond as the attractive force that holds atoms, ions, or molecules together. It emphasises that most atoms do not exist independently in nature (except noble gases) and tend to achieve stability by completing their valence shell.
A recurring idea throughout the chapter is the octet rule, which states that atoms combine by gaining, losing, or sharing electrons to achieve eight electrons in their outermost shell. The PDF also highlights the limitations of the octet rule, such as:
- Molecules with odd numbers of electrons (NO, NO₂)
- Expanded octet species (PF₅, SF₆)
- Electron-deficient molecules (BeCl₂, BCl₃)
Ionic Bonding and Its Characteristics
The PDF explains ionic (electrovalent) bonding as the bond formed due to electrostatic attraction between oppositely charged ions produced by electron transfer from a metal to a non-metal.
Key properties of ionic compounds mentioned include:
- High melting and boiling points
- Hard and brittle nature
- Conduct electricity in molten state or aqueous solution
- Non-directional bonds
- High lattice enthalpy
Examples such as NaCl and CaCl₂ are used to illustrate ionic bond formation.
Covalent Bonding and Its Features
A covalent bond is described as the sharing of electron pairs between atoms. Depending on the number of shared electrons, bonds are classified as:
- Single bond
- Double bond
- Triple bond
The PDF also introduces Lewis structures for covalent and ionic compounds and explains how they help visualise bonding.
Properties of covalent compounds highlighted include:
- Low melting and boiling points
- Poor electrical conductivity
- Solubility in non-polar solvents
- Slow reaction rates
Formal Charge and Bond Order
The concept of formal charge is explained using a formula based on valence electrons, lone pair electrons, and shared electrons. The PDF stresses that structures with minimum formal charges are usually more stable.
Bond order is defined as the number of bonds between two atoms. Important relationships mentioned:
- Higher bond order → shorter bond length
- Higher bond order → greater bond strength
Isoelectronic species with the same bond order are also discussed.
Polar and Non-Polar Covalent Bonds
The chapter distinguishes between:
- Non-polar covalent bonds (equal sharing, same atoms)
- Polar covalent bonds (unequal sharing, different atoms)
The concept of dipole moment is introduced as a measure of bond polarity and molecular polarity.
Download this CLASS 11 – CHEMICAL BONDING MOLECULAR STRUCTURE PDF File: Click Here
Resonance and Resonance Energy
The PDF explains that some molecules cannot be represented by a single Lewis structure. Instead, their actual structure is a resonance hybrid of multiple canonical forms.
Key points include:
- Conditions for writing resonance structures
- Resonance increases stability
- Resonance energy is the difference between actual bond energy and the most stable canonical structure
Examples like ozone and carbonate ion are discussed.
VSEPR Theory and Molecular Shapes
The Valence Shell Electron Pair Repulsion (VSEPR) Theory states that electron pairs around a central atom arrange themselves to minimise repulsion.
Important ideas covered:
- Shape depends on total number of bond pairs and lone pairs
- Repulsion order:
Lone pair–lone pair > Lone pair–bond pair > Bond pair–bond pair - Lone pairs distort ideal geometries
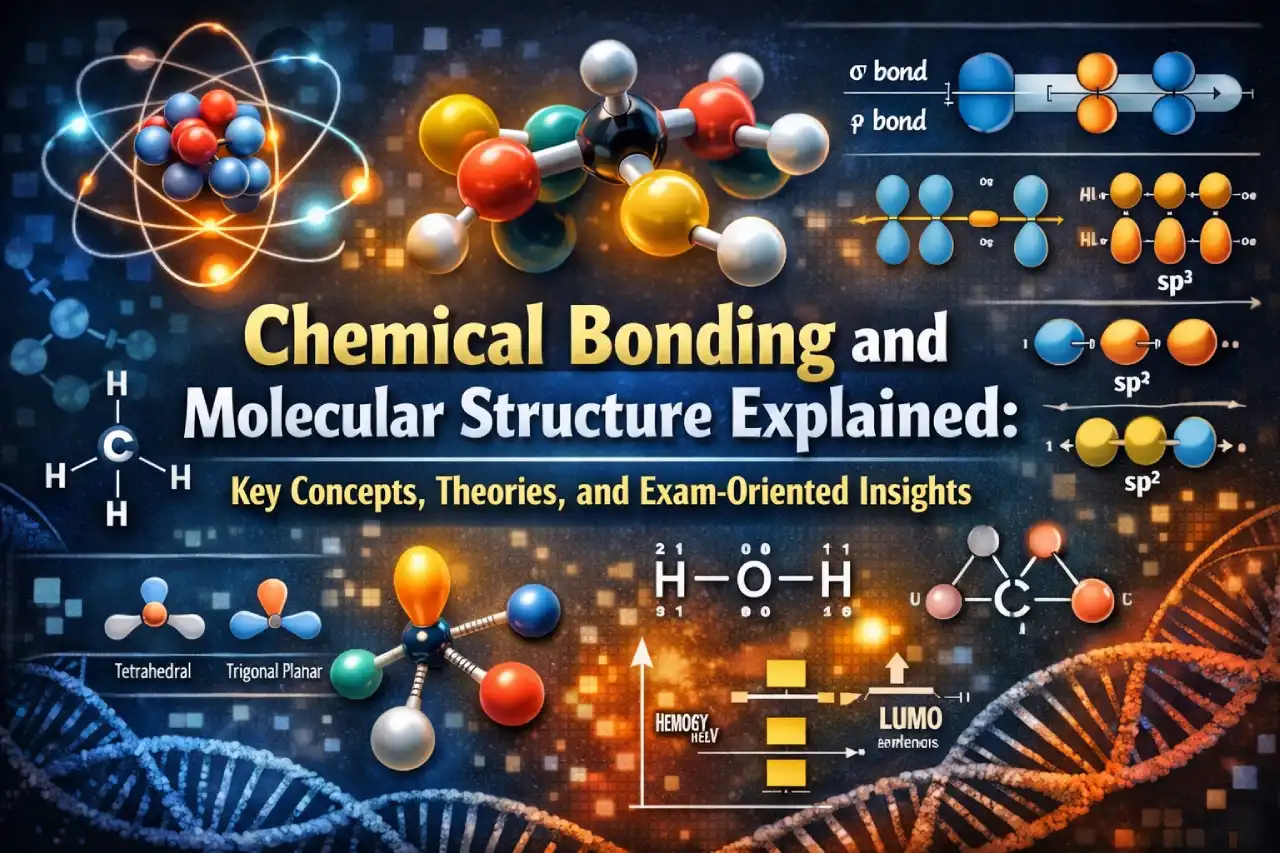
Examples include linear, bent, trigonal planar, tetrahedral, pyramidal, and angular molecules.
Valence Bond Theory (VBT)
According to VBT, a covalent bond forms by overlap of half-filled atomic orbitals. Greater overlap leads to stronger bonds.
The PDF explains two types of overlaps:
- Sigma (σ) bond – head-on overlap
- Pi (π) bond – sidewise overlap
Sigma bonds are stronger than pi bonds.
Hybridisation
Hybridisation is defined as intermixing of orbitals to form equivalent hybrid orbitals.
Types discussed:
- sp (linear, 180°)
- sp² (trigonal planar, 120°)
- sp³ (tetrahedral, 109°28’)
Examples such as BeCl₂, BF₃, CH₄, and CCl₄ are included.
Molecular Orbital Theory (MOT)
The PDF presents MOT as a theory where atomic orbitals combine to form molecular orbitals:
- Bonding molecular orbitals
- Anti-bonding molecular orbitals
- Non-bonding molecular orbitals
Bond order in MOT is given as:
Bond order = ½ (Number of bonding electrons − Number of antibonding electrons)
Magnetic behaviour is linked to electron pairing:
- All electrons paired → diamagnetic
- One or more unpaired electrons → paramagnetic
Hydrogen Bonding
Hydrogen bonding occurs when hydrogen attached to F, O, or N interacts with another electronegative atom.
Two types are explained:
- Intermolecular hydrogen bonding
- Intramolecular hydrogen bonding
Applications discussed include:
- High boiling point of water
- Lower density of ice compared to water
- Maximum density of water at 4°C
Practice Questions and Exam Orientation
The PDF contains:
- Multiple choice questions
- Assertion–reason questions
- Short answer and long answer questions
- Numerical and conceptual problems
Most questions are drawn from NCERT Exemplar, making the material exam-focused.