Electrochemistry is one of the most important chapters in senior secondary Chemistry, especially for Class 12 students preparing for board exams and competitive tests like NEET and JEE. This topic deals with the relationship between electrical energy and chemical reactions, explaining how chemical reactions can generate electricity and how electrical energy can drive chemical changes. The chapter introduces electrochemical cells, electrode potentials, conductance of electrolytes, electrolysis, batteries, fuel cells, and corrosion.
I am writing about this topic because electrochemistry often feels difficult to students due to the number of formulas, numerical problems, and concepts involved. However, once the basics are clear, this chapter becomes highly scoring. A strong understanding of electrochemistry also helps students connect Chemistry with real-life applications like batteries, electroplating, fuel cells, and corrosion prevention, which are all discussed in detail in the syllabus.
What Is Electrochemistry?
Electrochemistry is the branch of chemistry that studies the interconversion of chemical energy and electrical energy. It mainly focuses on redox reactions, where oxidation and reduction occur simultaneously. These reactions form the foundation of electrochemical cells.
In simple terms, electrochemistry explains:
- How chemical reactions produce electricity
- How electricity is used to cause chemical reactions
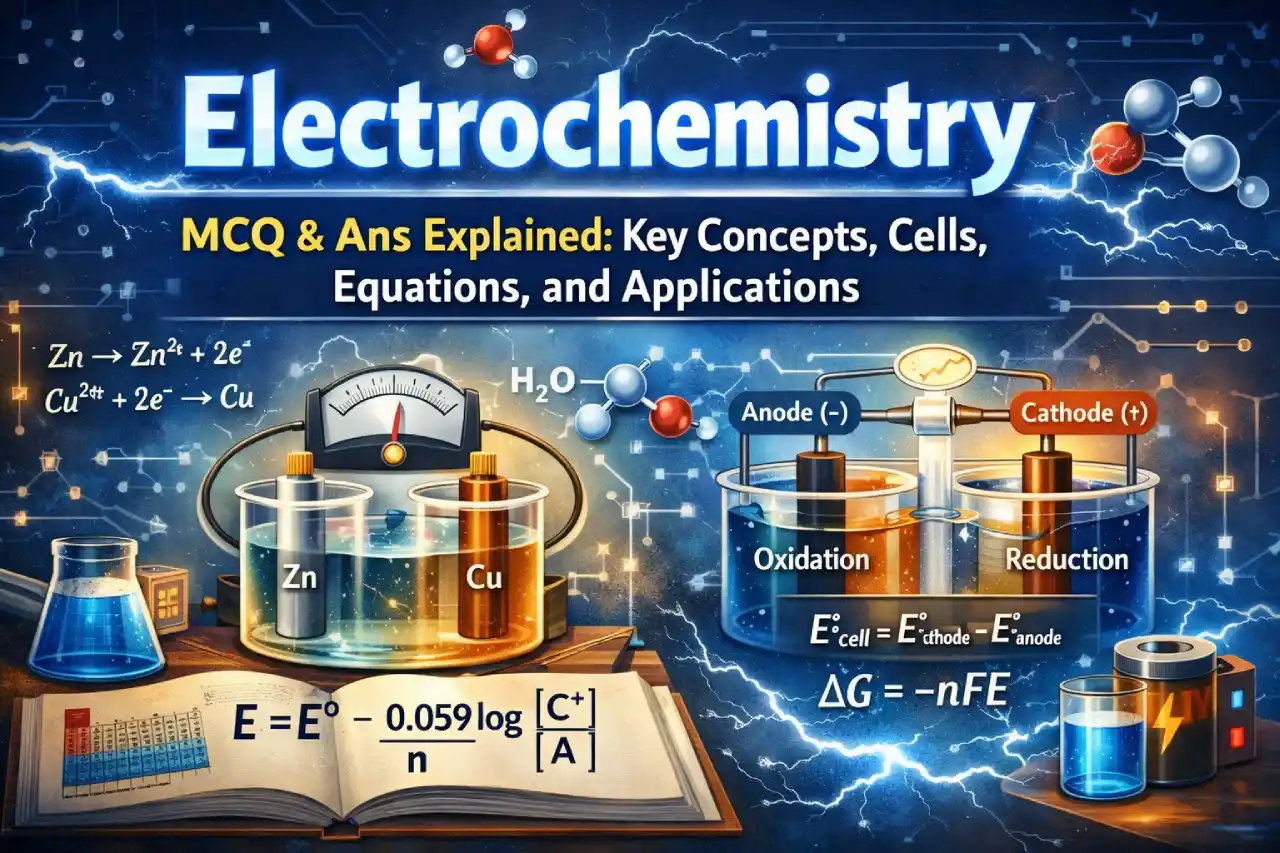
Electrochemical Cells and Their Types
An electrochemical cell is a device that converts chemical energy into electrical energy or vice versa. Based on how the energy conversion happens, electrochemical cells are classified into two main types.
Galvanic (Voltaic) Cells
Galvanic cells convert chemical energy into electrical energy through spontaneous redox reactions. A classic example is the Daniell cell, where zinc undergoes oxidation and copper undergoes reduction.
Key points covered in the PDF include:
- Oxidation occurs at the anode
- Reduction occurs at the cathode
- Electrons flow from anode to cathode through the external circuit
- A salt bridge is used to maintain electrical neutrality
Electrolytic Cells
Electrolytic cells use external electrical energy to drive non-spontaneous chemical reactions. These cells are commonly used in electrolysis, electroplating, and metal refining.
Important features include:
- Electrical energy is supplied from an external source
- Oxidation still occurs at the anode
- Reduction occurs at the cathode
- Used in industrial applications
Electrode Potential and Standard Electrode Potential
Electrode potential is the tendency of an electrode to lose or gain electrons when it is in contact with its ions in solution. It is measured relative to a standard hydrogen electrode.
Standard electrode potential:
- Is measured under standard conditions (1 M concentration, 1 atm pressure, 298 K temperature)
- Helps in predicting the direction of electron flow
- Determines oxidising and reducing strength of substances
Download this ELECTROCHEMISTRY PDF File: Click Here
Nernst Equation and Its Importance
The Nernst equation is used to calculate the electrode potential of a cell under non-standard conditions. This equation is extremely important for numerical problems.
The equation shows that electrode potential depends on:
- Concentration of ions
- Temperature
- Number of electrons transferred
The PDF includes multiple applications of the Nernst equation to calculate cell potential, equilibrium constant, and Gibbs free energy change.
Conductance of Electrolytic Solutions
This section explains how electricity is conducted through electrolyte solutions by the movement of ions.
Key concepts discussed include:
- Specific conductance
- Molar conductance
- Equivalent conductance
- Effect of dilution on conductance
Strong electrolytes show high conductance, while weak electrolytes show low conductance due to partial ionisation.
Electrolysis and Faraday’s Laws
Electrolysis is the process of using electrical energy to bring about chemical decomposition. Faraday’s laws of electrolysis explain the quantitative relationship between the amount of electricity passed and the mass of substance deposited or liberated.
According to the laws:
- The mass of substance deposited is directly proportional to the quantity of electricity passed
- Different substances require different amounts of charge depending on their equivalent weight
These principles are widely used in numerical problems and industrial applications.
Batteries and Fuel Cells
The chapter also covers different types of batteries such as dry cells, lead storage batteries, nickel-cadmium cells, and mercury cells.
Fuel cells, especially hydrogen-oxygen fuel cells, are highlighted as eco-friendly energy sources. They directly convert chemical energy into electrical energy with high efficiency and minimal pollution.
Corrosion and Its Prevention
Corrosion is the gradual deterioration of metals due to chemical or electrochemical reactions with the environment. Rusting of iron is a common example discussed in the chapter.
Methods of prevention include:
- Galvanisation
- Cathodic protection
- Protective coatings
Understanding corrosion is important not only for exams but also for real-world engineering and construction applications.